If you haven't experienced adequate results with medication and other conservative treatments, your surgeon may recommend knee replacement surgery, which may allow you to return to the everyday activities you enjoy.
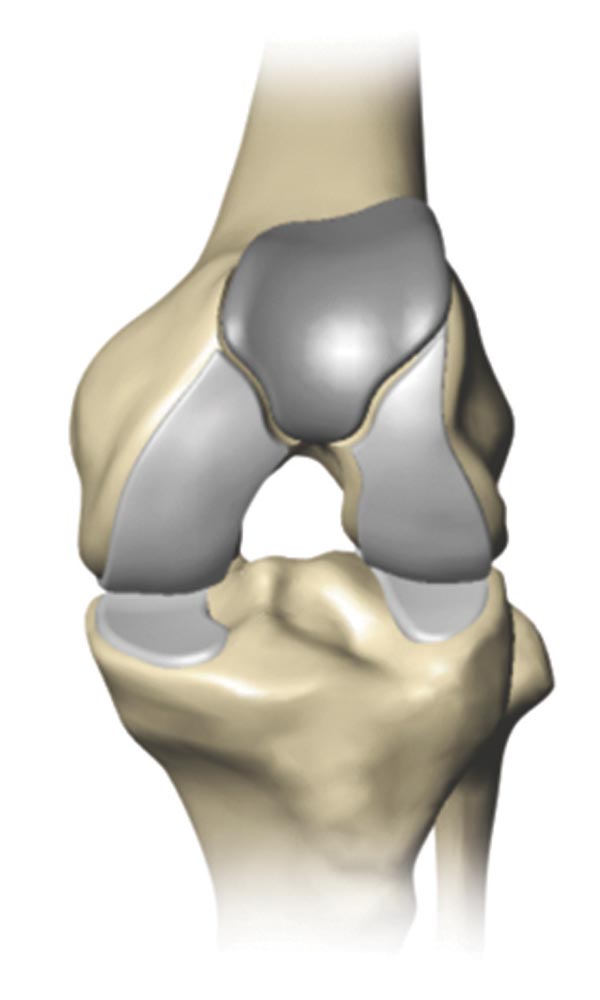
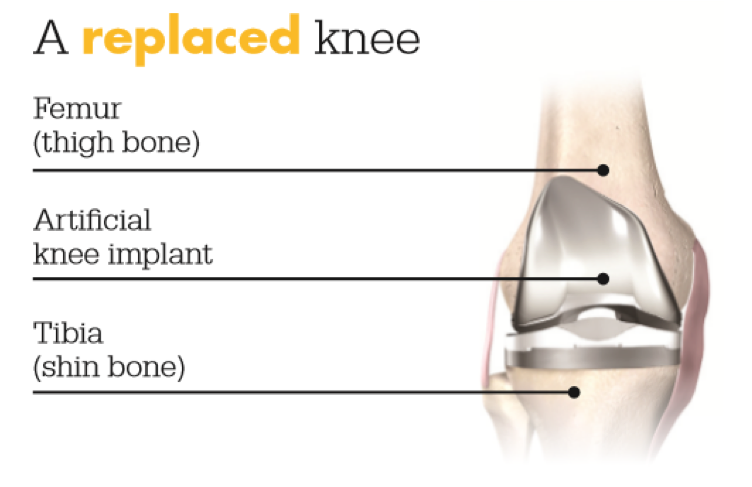
Knee replacement is a surgical procedure in which a diseased or damaged joint is replaced with an artificial joint called an implant.
Made of metal alloys and high grade plastics (to better match the function of bone and cartilage, respectively), the implant is designed to move much like a healthy human joint.
Over the years, knee replacement techniques and instrumentation have undergone countless improvements. Stryker’s Mako Robotic-Arm Assisted Technology is an example of how technology is transforming the way joint replacement surgeries are being performed.
Like any surgery, joint replacement surgery has serious risks.
Did you know?
Depending on each patient's conditions, realistic activities following knee replacement may include walking, biking, swimming and other low impact activities.1

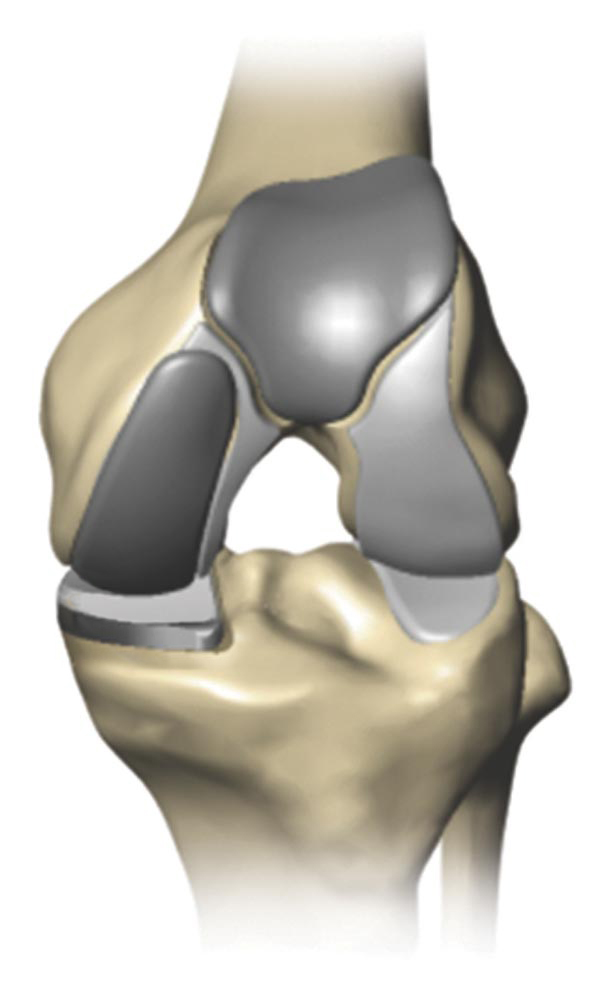
Total knee replacement is a surgical procedure in which a diseased or damaged joint is replaced with an artificial joint called an implant. Made of metal alloys and high grade plastics (to better match the function of bone and cartilage, respectively), the implant is designed to move much like a healthy human joint.
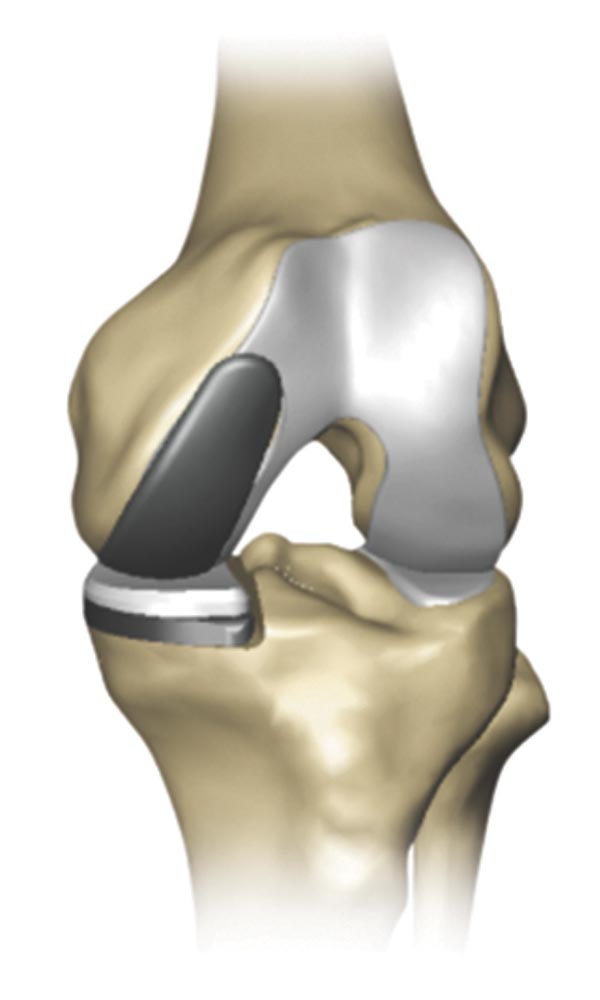
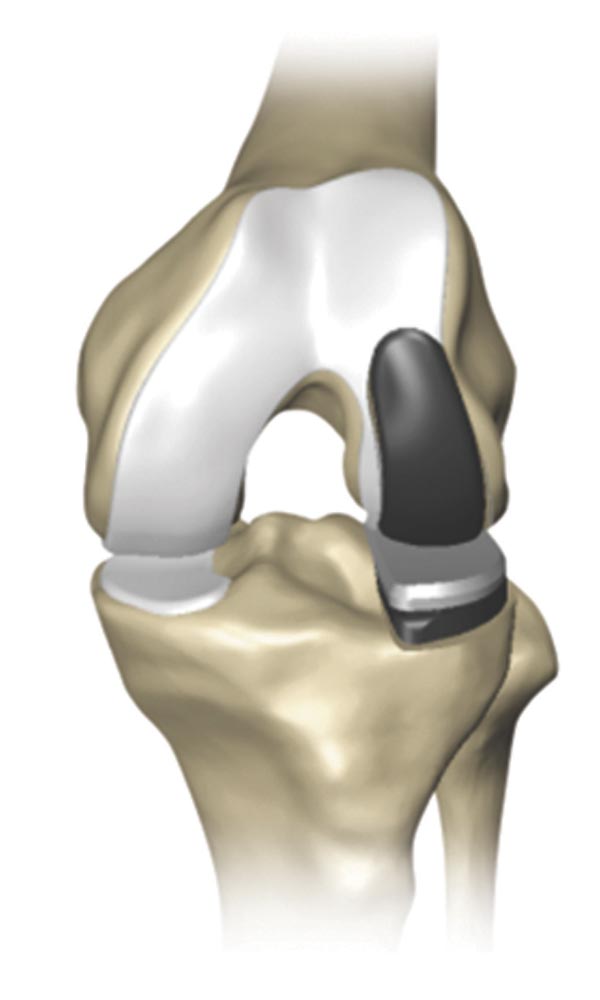
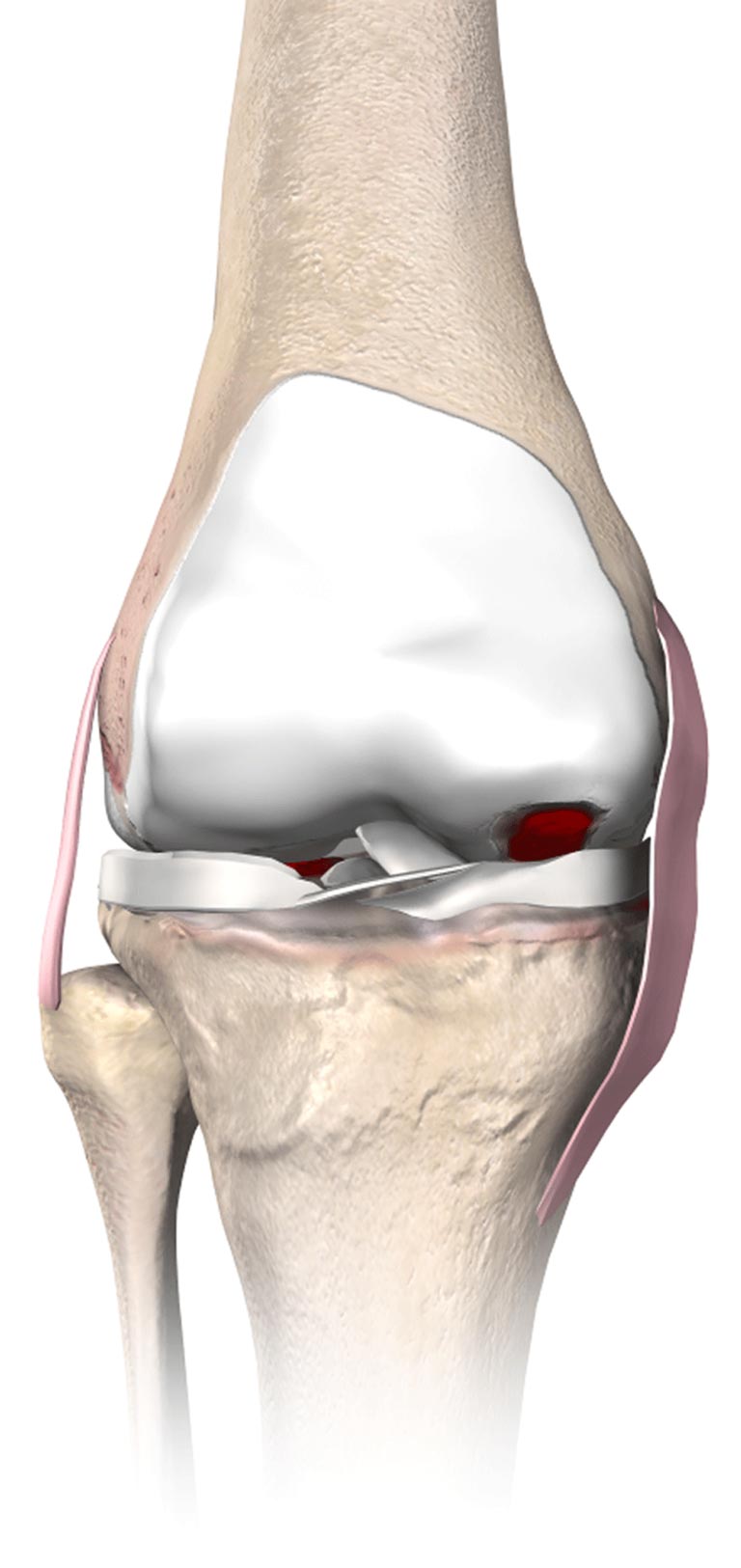
Partial knee replacement (PKR) is a surgical procedure that helps relieve arthritis in one or two of the three compartments of the knee.
With PKR, only the damaged area of the knee joint is replaced, which may help to minimize trauma to healthy bone and tissue.2
Stryker has worked with surgeons to develop innovative products to be utilized in Partial Knee Replacement. Stryker’s Mako robotic-arm assisted technology can be used for partial knee replacement, which is a procedure designed to relieve the pain caused by joint degeneration due to osteoarthritis (OA).
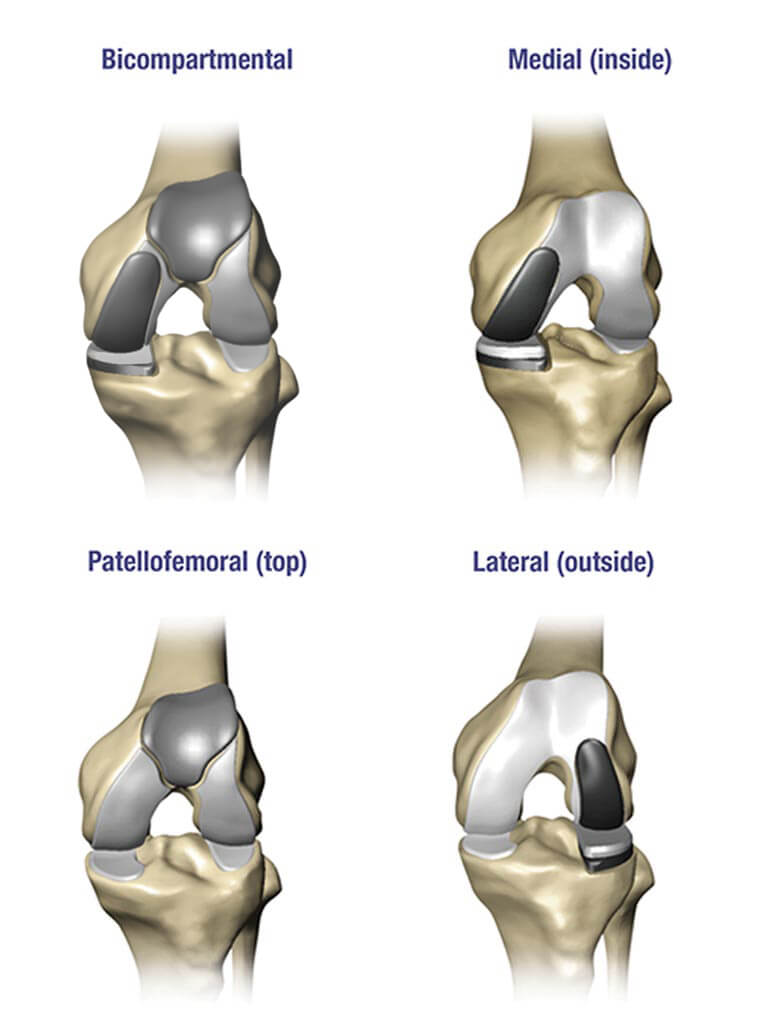
There are three types of Partial Knee Replacement.
Did you know?
Studies indicate that patients who undergo knee replacement may return to driving in four to six weeks3*
*Depending on each patient's conditions
IMPORTANT INFORMATION
Disclaimer
Hip joint replacement is intended for use in individuals with joint disease resulting from degenerative and rheumatoid arthritis, avascular necrosis, fracture of the neck of the femur or functional deformity of the hip.
Knee joint replacement is intended for use in individuals with joint disease resulting from degenerative, rheumatoid and post-traumatic arthritis, and for moderate deformity of the knee. Joint replacement surgery is not appropriate for patients with certain types of infections, any mental or neuromuscular disorder which would create an unacceptable risk of prosthesis instability, prosthesis fixation failure or complications in postoperative care, compromised bone stock, skeletal immaturity, severe instability of the joint, or excessive body weight.
Like any surgery, joint replacement surgery has serious risks which include, but are not limited to, pain, bone fracture, change in the treated leg length (hip), joint stiffness, hip joint fusion, amputation, peripheral neuropathies (nerve damage), circulatory compromise (including deep vein thrombosis (blood clots in the legs)), genitourinary disorders (including kidney failure), gastrointestinal disorders (including paralytic ileus (loss of intestinal digestive movement)), vascular disorders (including thrombus (blood clots), blood loss, or changes in blood pressure or heart rhythm), bronchopulmonary disorders (including emboli, stroke or pneumonia), heart attack, and death.
Implant related risks which may lead to a revision of the implant include dislocation, loosening, fracture, nerve damage, heterotopic bone formation (abnormal bone growth in tissue), wear of the implant, metal sensitivity, soft tissue imbalance, osteolysis (localized progressive bone loss), audible sounds during motion, and reaction to particle debris.
The information presented is for educational purposes only. Speak to your doctor to which therapy is appropriate for you. Individual results vary and not all patients will return to the same activity level. The lifetime of any joint replacement is limited and depends on several factors like patient weight and activity level. Your doctor will counsel you about strategies to potentially prolong the lifetime of the device, including avoiding high-impact activities, such as running, as well as maintaining a healthy weight. It is important to closely follow your physician’s instructions regarding post-surgery activity, treatment and follow-up care.
Speak to your doctor to decide which therapy/treatment is appropriate for you.
Stryker Corporation or its other divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: Mako, Stryker, Together with our customers, we are driven to make healthcare better. All other trademarks are trademarks of their respective owners or holders.
GSNPS-PE-85_17642
REFERENCES
1. Foran, Jared R.H., MD. Total knee replacement. American Academy of Orthopaedic Surgeons. (2015) Retrieved February 2017. http://orthoinfo.aaos.org/topic. cfm?topic=A00389.
2. Arno, S; Maffei, D; Walker, PS; Schwartzkopf, R; Desai, P; Steiner, GC. Retrospective Analysis of Total Knee Arthroplasty Cases for Visual, Histological and Clinical Eligibility of Unicompartmental Knee Arthroplasties. J. Arthroplasty. 2011. 26(8): 1396-1403.
3. Marecek GS, Schafer MF. Driving after orthopaedic surgery. J Am Acad Orthop Surg. 2013 Nov;21(11):696-706
Please upgrade your internet browser.
Our website was designed for a range of browsers. However, if you would like to use many of our latest and greatest features, please upgrade to a modern, fully supported browser.
Find the latest versions of modern supported browsers »Note: If you are browsing in Internet Explorer 9 or later, and you are still seeing this message, you may be in Compatability Mode. Look for the Compability View icon in your Address bar, or the Compatability View and Compatability View settings from the Tools Menu in your Internet Explorer toolbar (hit the ALT key if your menu bar is hidden). If you are on an Intranet, you may need to contact your IT Support about Compatability View settings and whitelists.